
Cauvery College
of Pharmacy
Dr. Chandrashekar Foundation(R)
Pharmaceutics
Department of Pharmaceutics
The Department of Pharmaceutics is a core pillar of pharmacy education, focused on the science of transforming a drug substance into a safe and effective medication. It bridges the gap between pure chemistry and clinical application by focusing on dosage form design and drug delivery systems.
Pharmaceutics
It is the specialized science of dosage form design, focusing on turning a pure drug chemical into a safe and effective medication for patients. It covers the entire lifecycle of a medicine, from formulation development and large-scale manufacturing to quality control and packaging.
The field integrates physical pharmacy and technology to create various delivery systems like tablets, injections, ointments, and advanced nanoparticles. A major goal is optimizing drug delivery, ensuring the active ingredient is stable and released at the right site in the body for maximum effect.

Pharmaceutical Engineering
Pharmaceutical Engineering focuses on the design, operation, and optimization of unit operations – like milling, mixing, and filtration – essential for large-scale drug production. It applies principles of chemical engineering to ensure the efficient transfer of heat, mass, and energy during the manufacturing of various dosage forms.
The field emphasizes the selection and maintenance of industrial machinery, such as rotary tablet presses and fluid bed dryers, to maintain consistent product quality.

Industrial Pharmaceutics
Industrial Pharmacy is a specialized branch of the Department of Pharmaceutics that focuses on the large-scale manufacturing, development, and quality management of drug products. It bridges the gap between laboratory-scale drug discovery and mass commercial production.
Key Area of Study
The curriculum is typically split into foundational (Industrial Pharmacy I) and advanced (Industrial Pharmacy II)
• Dosage Form Manufacturing: Techniques for producing tablets, capsules, liquid orals, parenterals (injections), and cosmetics on an industrial scale
• Pilot Plant & Scale-Up: Learning how to move a successful lab formulation to a small pilot plant and eventually to a full-scale factory batch
• Packaging Technology: Selecting and testing materials like glass, plastic, and metal for drug stability and safety
Quality & Regulatory Framework
• Good Manufacturing Practices (GMP): Global standards ensured by bodies like the WHO and USFDA to ensure product consistency
• Quality Assurance (QA) & Quality Control (QC): QC focuses on testing the final product (purity, potency), while QA involves systemic processes to prevent defects during production
Specialized Laboratories
• Machine Room: Contains heavy equipment like rotary tablet presses, rapid mixer granulators, and fluid bed dryers
• Sterile Area: A highly controlled environment with Laminar Air Flow (LAF) for the manufacturing of eye drops and injectables

Bio - Pharmaceutics and Pharmacokinetics
Biopharmaceutics examines how a drug’s physical properties and dosage form design influence its rate of systemic absorption. Pharmacokinetics (PK) mathematically tracks “what the body does to the drug” through the ADME process: Absorption, Distribution, Metabolism, and Excretion.
It focuses on critical parameters like bioavailability, which measures the fraction of the dose that reaches the bloodstream unchanged. Scientists use PK modeling to calculate the half-life and clearance of a drug to determine the safest, most effective dosing schedule.
Together, these fields ensure that generic medications are bioequivalent to brand-name drugs, performing identically in the human body.
Novel Drug Delivery System
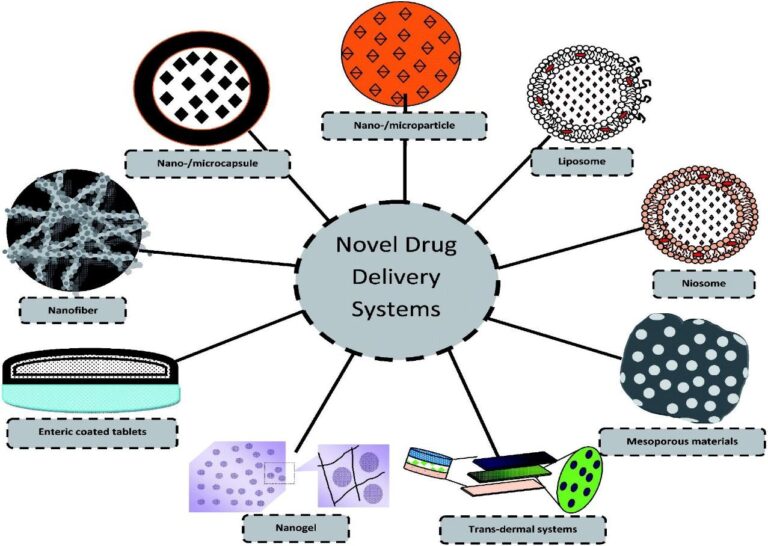
Novel Drug Delivery Systems (NDDS) represent advanced approaches that optimize the delivery of therapeutic agents to specific targeted sites in the body while minimizing side effects.
Unlike conventional dosage forms, NDDS uses specialized carriers like nanoparticles, liposomes, and niosomes to control the rate and timing of drug release. The primary goal is to maintain a constant therapeutic concentration in the blood, reducing the frequency of dosing and improving patient compliance.
These systems utilize advanced technologies such as transdermal patches, implants, and smart polymers that respond to biological triggers like pH or temperature.

Cosmetics
Cosmetics in pharmacy involves the science of formulating products like creams, lotions, and powders intended to enhance appearance or cleanse the skin, hair, and nails.
The field focuses on the physicochemical properties of ingredients to ensure they are stable, non-irritating, and aesthetically pleasing for the consumer. A key area is cosmeceuticals, which are cosmetic products containing biologically active ingredients that provide medicinal or drug-like benefits to the skin.
Our Faculty

Dr. R. Srinivasan
Principal
E-mail: principalccp570028@gmail.com
Qualification : MPharm, PhD.
Total experience 29 years
Publication 48
Book chapter 01

Dr. Jyothi S L
Professor and Head
E-mail: Jyothi.sana1320@gmail.com
Qualification: M. Pharm., PhD
No of Publications: 8
No of Patents: 2

Mrs. Shravya Lakshmi S
Associate Professor
E-mail: shravyals1993@gmail.com
Qualification: M. Pharm.
No of Publications: 11

Mrs. Rashmitha N M
Assistant Professor
E-mail: rashmithanm@gmail.com
Qualification: M. Pharm.
No of Publications: 03

Mr. Sunil Kumar B G
Associate Professor
E-mail: pharmasunilkumarbg@gmail.com
Qualification: M. Pharm.
No of Publications: 01

Mr. Chandrashekar PN
Assistant Professor
E-mail: chandru95pn@gmail.com
Qualification: M. Pharm.

Mr. Manishdev M
Assistant Professor
E-mail: mdgowda19@gmail.com
Qualification: M. Pharm.
No of Publications: 02

Ms. Roopashree GL
Assistant Professor
E-mail: roopashreegl16@gmail.com
Qualification: M. Pharma.
No of Publications: 01

Mrs. Ayesha Noorain KM
Assistant Professor
E-mail: aayeshanoorain@gmail.com
Qualification: M. Pharm.
No of Publications: 02

Mrs. Ranjini D S
Assistant Professor
E-mail: ranjukiran75@gmail.com
Qualification: M. Pharm.
No of Publications: 03